Plumeria Seedling Nutrition
Many people confuse plant nutrition with plant fertilization. Plant nutrition refers to the needs and uses of the basic chemical elements in the plant. Fertilization is the term used when these materials are supplied to the environment around the plant. A lot must happen before a chemical element supplied in a fertilizer can be taken up and used by the plant.
Foliar Absorption: Under normal growing conditions, plants absorb most nutrients, except carbon, hydrogen, and oxygen, from the soil. However, some nutrients can also be absorbed by the leaves if they are sprayed with a dilute solution. The factors that affect absorption by the cell are still important because the nutrient must enter the cell to be used by the plant. Care must be taken that the concentration of the nutrient is not too high or the leaf will be injured. Also, the leaf is covered by a thin layer of wax called the cuticle that the nutrient must get around or through before it can enter the cell.
Plumeria need 17 elements for normal growth. Carbon, hydrogen, and oxygen are found in air and water.
Nitrogen, potassium, magnesium, calcium, phosphorus, and sulfur are found in the soil and are used in relatively large amounts by the plant and are called macronutrients. There are eight other elements that are used in much smaller amounts; these are called micronutrients or trace elements. The micronutrients, which are found in the soil, are iron, zinc, molybdenum, manganese, boron, copper, cobalt, and chlorine. All 17 elements, both macronutrients and micronutrients, are essential for plant growth.
Fertilizer Nutrients
Plumeria need to be fertilized because most soil does not provide the essential nutrients required for optimum growth. Even if you are lucky enough to start with a great soil mix, as your plumeria grow, they absorb nutrients and leave the soil less fertile. Remember those beautiful blooms and leaves you grew last year? It took nutrients from the soil to build those plant tissues. By fertilizing your plumeria, you replenish lost nutrients and ensure that this year’s plumeria have the food they need to flourish.
There are six primary nutrients that plants require. Plants get the first three (carbon, hydrogen and oxygen) from air and water. The other three are nitrogen, phosphorus and potassium.
Nitrogen helps plumeria make the proteins they need to produce new tissues. In nature, nitrogen is often in short supply so plumeria have evolved to take up as much nitrogen as possible, even if it means not taking up other necessary elements. If too much nitrogen is available, the plumeria may grow abundant foliage but not produce flowers or may grow tall and lanky. Growth could be stunted because the plumeria isn’t absorbing enough of the other elements it needs.
Phosphorus stimulates root growth, helps the plant set buds and flowers, improves vitality and increases seed size. It does this by helping transfer energy from one part of the plumeria to another. To absorb phosphorus, most plumeria require a soil pH of approximately 6.5 to 6.8. Organic matter and the activity of soil organisms also increase the availability of phosphorus. Plumeria only require a small amount of Phosphorus, using high phosphorus fertilizers when you don’t need it could cause serious problems.
The buildup of phosphorus can cause plumeria to grow poorly and even die. Excessive soil phosphorus reduces the plant’s ability to take up required micronutrients, particularly iron and zinc, even when soil tests show there are adequate amounts of those nutrients in the soil.
Phosphorus buildup is caused by excessive use of inorganic fertilizer or the use of composts and manures high in phosphorus. High soil phosphorus levels also can threaten streams, rivers, lakes and oceans.
There are three additional nutrients that plumeria need, but in much smaller amounts:
Potassium: Unlike nitrogen and phosphorus, potassium is not used in the structural synthesis of bio-chemically important molecules. Potassium is found within the plant cell solution and is used for maintaining the turgor pressure of the cell (meaning it keeps the plant from wilting). In addition, potassium plays a role in the proper functioning of stomata (cells located on the bottom of the leaf that open and close to allow water vapor and waste gases to escape) and acts as an enzyme activator.
Calcium is used by plumeria in cell membranes, at their growing points and to neutralize toxic materials. Calcium, in the form of calcium pectate, is responsible for holding together the cell walls of plants. In addition, calcium improves soil structure and helps bind organic and inorganic particles together. When calcium is deficient, new tissue such as root tips, young leaves, and shoot tips often exhibit distorted growth from improper cell wall formation. Calcium is also used in activating certain enzymes and to send signals that coordinate certain cellular activities.
Magnesium is necessary to both plumeria growth and health. It is involved in several different processes, including photosynthesis, which nearly all living organisms are dependent on. It is the only metallic component of chlorophyll. Without it, plumeria can’t process sunlight.Magnesium (Mg), along with calcium and sulfur, is one of the three secondary nutrients required by plants for normal, healthy growth. Don’t be confused by the term “secondary” as it refers to the quantity and not the importance of a nutrient. A lack of a secondary nutrient is just as detrimental to plant growth as a deficiency of any one of the three primary nutrients (nitrogen, phosphorus and potassium) or a deficiency of micronutrients (iron, manganese, boron, zinc, copper and molybdenum). Furthermore, in some plants, the tissue concentration of magnesium is comparable to that of phosphorus, a primary nutrient.
The most important role of magnesium is as the central atom in the chlorophyll molecule. Chlorophyll is the pigment that gives plants their green color and carries out the process of photosynthesis. It also aids in the activation of many plant enzymes needed for growth and contributes to protein synthesis.
Sulphur along with calcium and magnesium, is one of the three secondary nutrients required by plumeria for normal, healthy growth. A deficiency in a secondary nutrient is just as detrimental as a deficiency in nitrogen, phosphorus or potassium. Sulfur is often overlooked and underappreciated in importance. There is a significant balance between nitrogen and sulfur. Without enough sulfur, plumeria cannot efficiently use nitrogen and other nutrients to reach their full potential.
Plants acquire sulfur from the growing medium as sulfate. Sulfate is readily soluble and subject to loss by leaching. Plumeria metabolism reduces sulfate and sulfur dioxide to forms that can be used to build organic molecules. Sulfur is a vital part of all plant proteins, and certain plant hormones. It is also used in the formation of certain oils and volatile compounds found in the onion and garlic family.
Finally, there are eight elements that plumeria need in tiny amounts. These are called micronutrients and include boron, chlorine, copper, cobalt , iron, manganese, molybdenum, and zinc.
Healthy soil that is high in organic matter usually contains adequate amounts of each of these micronutrients.
Organic vs. Synthetic
Do plumeria really care where they get their nutrients? Yes, because organic and synthetic fertilizers provide nutrients in different ways. Organic fertilizers are made from naturally occurring mineral deposits and organic material, such as bone or plant meal or composted manure. Synthetic fertilizers are made by chemically processing raw materials.
In general, the nutrients in organic fertilizers are not water-soluble and are released to the plumeria slowly over a period of months or even years. For this reason, organic fertilizers are best applied in the fall so the nutrients will be available in the spring. These organic fertilizers stimulate beneficial soil microorganisms and improve the structure of the soil. Soil microbes play an important role in converting organic fertilizers into soluble nutrients that can be absorbed by your plumeria. In most cases, organic fertilizers and compost will provide all the secondary and micronutrients your plumeria need.
Synthetic fertilizers are water-soluble and can be taken up by the plumeria almost immediately. In fact applying too much synthetic fertilizer can “burn” foliage and damage your plumeria. Synthetic fertilizers give plumeria a quick boost but do little to improve soil texture, stimulate soil life, or improve your soil’s long-term fertility. Because synthetic fertilizers are highly water-soluble, they can also leach out into streams and ponds. Synthetic fertilizers do have some advantages in early spring. Because they are water-soluble, they are available to plumeria even when the soil is still cold and soil microbes are inactive. For this reason, some organically-based fertilizers, such as PHC All-Purpose Fertilizer, also contain small amounts of synthetic fertilizers to ensure the availability of nutrients.
For the long-term health of your garden, feeding your plumeria by building the soil with organic fertilizers and compost is best. This will give you soil that is rich in organic matter and teeming with microbial life.
Foliar Feeding?
Plumeria can absorb nutrients eight to 20 times more efficiently through their leaf surfaces than through their roots. As a result, spraying foliage with liquid nutrients can produce remarkable yields. For best results, spray plants during their critical growth stages such as transplanting time and blooming time.
What About pH?
Even if proper nutrients are present in the soil, some nutrients cannot be absorbed by plumeria if the soil pH is too high or too low. For most plumeria, soil pH should be between 6.0 and 7.0. A soil test will measure the pH of your soil. You can send a sample to a lab (contact your local extension service for a low-cost kit) or buy a home kit and do it yourself. Lime or wood ash can be used to raise pH; sulfur or aluminum sulfate can lower pH. Keep in mind that it’s best to raise or lower soil pH slowly over the course of a year or two. Dramatic adjustments can result in the opposite extreme, which may be worse than what you started with. Once again, a helpful solution is to apply compost. Compost moderates soil pH and is one of the best ways to maintain the 6.5 ideal.
Slow-release, granular Excalibur 11-11-13 or similar fertilizer gives your plumeria all the nutrients they need, including plenty of phosphorus for big, abundant flowers. For a healthy start, mix a handful into the soil at transplant time and at the beginning of your growing season.
How to Choose a Fertilizer
In most cases, an all-purpose, 11-11-13 fertilizer with micronutrients such as Excalibur will provide the nutrients all plumeria need for healthy growth. If a soil test reveals certain nutrient deficiencies, or if you want to tailor your fertilizer to the needs of particular plumeria, you can select a special formulation. What you choose will depend on your soil and what you are growing.
The three numbers that you see on a fertilizer label, such as 11-11-13, tell you what proportion of each macronutrient the fertilizer contains. The first number is always nitrogen (N), the second is phosphorus (P) and the third is potassium (K). This “N-P-K” ratio reflects the available nutrients —by weight—contained in that fertilizer. For example, if a 100-pound bag of fertilizer has an N-P-K ratio of 11-11-13, it contains 11 pounds of nitrate, 11 pounds of phosphate (which contains phosphorus), 13 pounds of potash (which contains potassium) and 84 pounds of filler.
Note that the N-P-K ratio of organic fertilizers is typically lower than that of a synthetic fertilizer. This is because by law, the ratio can only express nutrients that are immediately available. Most organic fertilizers contain slow-release nutrients that will become available over time. They also contain many trace elements that might not be supplied by synthetic fertilizers.
To build the long-term health and fertility of your soil, we recommend using granular slow release fertilizers with micronutrients. Supplemented with a water-soluble fertilizer ensures that your plants have the nutrients they need when they’re in active growth.

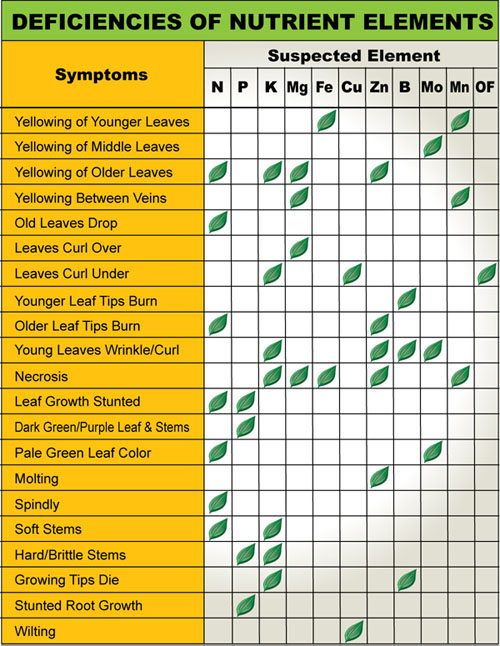
 After trying every fertilizer you can think of and searching for Fertilizers specifically formulated for Plumeria and not finding anything worth using, we decided we had to have a professional company custom mix a fertilizer. We knew from years of experience that Plumeria do best when you feed them a balanced fertilizer that provides nutrients as Plumeria need them. We believe that healthier plants will result in better growing habits and more blooms.
After trying every fertilizer you can think of and searching for Fertilizers specifically formulated for Plumeria and not finding anything worth using, we decided we had to have a professional company custom mix a fertilizer. We knew from years of experience that Plumeria do best when you feed them a balanced fertilizer that provides nutrients as Plumeria need them. We believe that healthier plants will result in better growing habits and more blooms.